

|
RESEARCH |
|
AWARDS |
|
PUBLICATIONS |
|
PRESENTATIONS |
|
TEACHING |
|
EMPLOYMENT |
|
SERVICE |
|
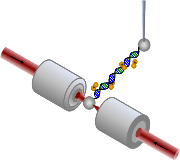
My research interest is in the field of biophysics. Biophysics is an interdisciplinary field, in which physics techniques are used to study biological systems. My specialty is the sub-division of biophysics known as single molecule biophysics where the interactions of biological systems are studied at the molecular level by isolating individual molecules. Optical Tweezers are one of the popular techniques that use lasers to isolate single biomolecules. In fact, the 2018 Nobel Prize in physics was awarded to “Optical Tweezers and their application to Biological Systems.” We have custom-built dual beam optical tweezers at Bridgewater State University, which use two counter-propagating laser beams to trap a single DNA molecule inside a flow chamber. We add anticancer drugs, small molecules that are potential cancer drugs, and proteins into the flow chamber to study their interactions with DNA at the single molecule level. |
|
Threading Ruthenium Complexes
Transition metal-based complexes have been successful in cancer treatment. This led to the exploration of Ruthenium based small molecules. Since Ruthenium based complexes have entered clinical trials for cancer therapy, several modifications have been made to make them more effective. One of these modifications is to covalently link two mononuclear ruthenium complexes to form binuclear complexes which were first developed at Chalmers University of Technology, Sweden. The slow kinetics of these molecules made it hard to study in bulk experiments, but by mechanically manipulating the DNA molecule using optical tweezers we explored the threading of these binuclear ruthenium complexes. This project is carried out in collaboration with Williams Lab at Northeastern University and Westerlund Lab at Chalmers University of technology, Sweden. The results of this project are being published in the Journal of American Chemical Society (2008), Nucleic Acids Research (2014) and Biochemistry (2018). Currently we are currently exploring how the chirality of the side chains of these threading intercalators affect the binding of them to the DNA.
Doxorubicin & Nogalamycin
We are also studying the interactions of two popular cancer drugs to characterize their binding properties. Nogalamycin has been used for a long time to treat cancer and was recently discontinued. On the other hand, Doxorubicin is one of the popular drugs in the market. Studying these two at the single molecule level and comparing will provide insights to develop better drug designs in the future. |
|
Many Single Molecules (using TIRF)
The other component of my research is conducted at Brandeis in collaboration with Prof. Jeff Gelles and Prof. Jane Kodev. We are exploring the pathways of oligonucleotide dissociations using Total Internal Reflection Fluorescence (TIRF) microscope. Fluorescently labeled receptors and ligands are imaged as a collection of single molecules by exciting them with multi-wavelength lasers to watch the kinetics. We explore the effect of rapid rebinding to explain the observations reported recently from several labs where the concentration dependent dissociation of biological molecules in relatively crowded environment. |


|
INTRODUCTION |


|
RESEARCH INTEREST |

|
T H A Y A P A R A M A N A T H A N |
|
WILLIAMS LAB |
|
GELLES LAB |
|
BSU-SMB LAB |
|
BSU PHYSICS |